The C-H bond is one of the most difficult to activate bonds in organic chemistry. The selective activation of inert C-H bonds are of strong interest for the preparation of basic and fine chemicals from natural oil, gas and coal resources and for the synthesis of complex agents, e. g. pharmaceutical products. Nature frequently uses dinuclear metallo-enzymes for selective C-H activations. The metal ions (mainly Fe and Cu) in the active sites of these enzymes store the oxidation and / or oxygenation equivalents of dioxygen during catalytic cycles. Examples for copper containing enzymes are tyrosinase and particulate methane monooxygenase, while soluble methane monooxygenase (sMMO) is the most prominent example for an iron containing enzyme for C-H activation. However, there is a growing class of non-heme diiron enzymes (NHFe2), which enable a breadth of divergent reactivities in natural product biosynthesis that emanate from a similar diiron core.
In general, the reduced FeIIFeII form reacts with O2 to yield a m-1,2-peroxo FeIIIFeIII intermediate (P), which is in most NHFe₂ enzymes the catalytically active species. In some cases, P intermediates undergo a proton-promoted activation step to yield P¢-type intermediates, for which FeIIIFeIII with m-1,1-peroxo, m-1,1-, or m-1,2-hydroperoxo ligands have been proposed. In sMMO, intermediate P converts in an also proton-promoted step to a high-valent FeIVFeIV active species (Q), which is generally believed to be the active intermediate for methane oxidation.
In order to develop better oxidation catalysts inspired by biology, we have developed a bis(tetradentate) dinucleating ligand system to generate dinuclear peroxo and high-valent species with a special focus on the correlation of their stabilities and reactivities as a function of the terminal donors.
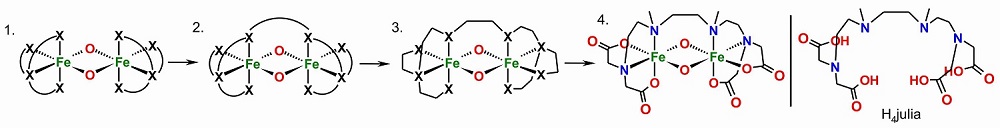
The terminal donors ranges from carboxylates, phenolates, imidazoles, to differently substituted pyridines:
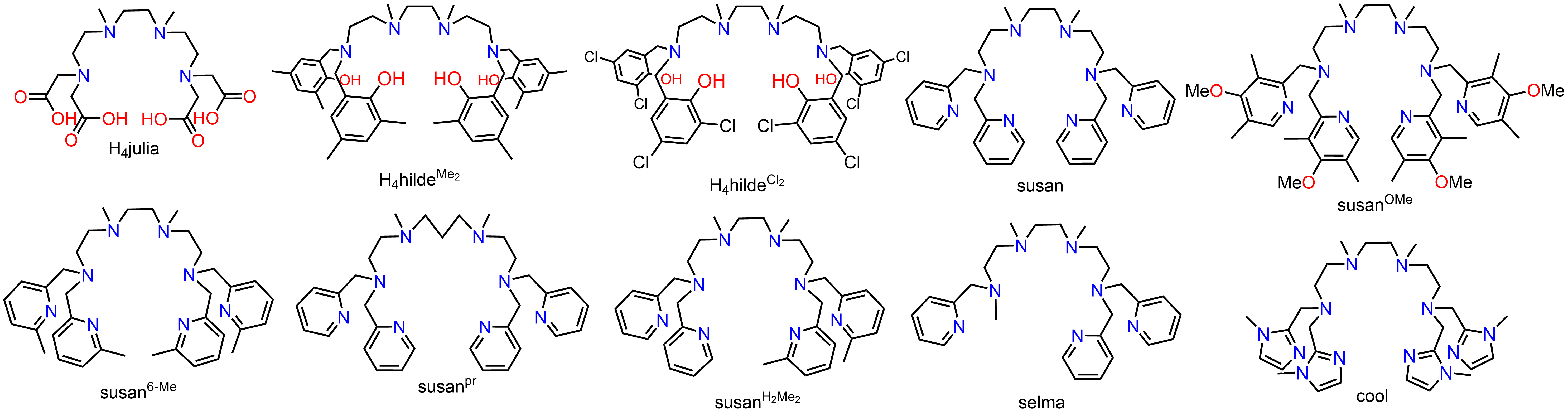
The ligand susan6-Me stabilizes the µ-1,2-peroxo complex [(susan6-Me){FeIII(µ-1,2-peroxo)(µ-O)FeIII}](ClO4)2, which was obtained from the reaction of [(susan6-Me){FeII(µ-OH)2FeII}]2+ with O2 and characterized in the solid state including sc-XRD and magnetism as well as in solution spectroscopically and electrochemically. The reversible oxidation provides the unprecedented high-valent peroxo complex [(susan6-Me){FeIV(µ-1,2-peroxo)(µ-O)FeIII}]3+ with a FeIV ion in local high spin state (Si = 2) and the reversible protonation to the also unprecedented µ-1,2-hydroperoxo complex [(susan6-Me){FeIII(µ-1,2-OOH)(µ-O)FeIII}]3+ as a model for P'-type intermediates.

In contrast, the µ-1,2-peroxo complex of susan is not stable but therefore reactive in HAT and OAT and is the first peroxo diferric model complexes showing oxidative reac-tivity as in the enzymes

Related Publications
- S. Jansen, F. L. B. Röhs, T. P. Zimmermann, S. Walleck, M. van Gastel, A. Tamhankar, B. Zhang, S. A. V. Jannuzzi, I. Kellner, I. Ivanovic-Burmazovic, S. DeBeer; and T. Glaser
"Formation Pathways of an Electrophilic µ-1,2-Peroxo FeIIIFeIII Complex: Spectroscopic Characterization and Reactivity"
J. Am. Chem. Soc., 2026, 148, 7065-7083
- S. Jansen, S. Walleck, A. Stammler, H. Bögge, and T. Glaser
"Influence of Asymmetry on the Molecular and Electronic Structures of µ-Oxo Diferric Complexes"
Z. Anorg. Allg. Chem., 2026 , 652, e202500176 - F. Depenbrock, E. Bill, M. van Gastel, D. J. SantaLucia, S. Walleck, J. Oldengott, A. Stammler, and T. Glaser
"Activation of Electrophilic Reactivity by Protonation and Oxidation of a Peroxo CoIIICoIII Complex: Reactivity and Molecular Structures of Hydroperoxo, Superoxo, and Peroxo CoIIICoIII Complexes"
J. Am. Chem. Soc., 2025, 147, 5775-5784
- L. Siebe, C. Butenuth, A. Köhler, A. Stammler, J. Oldengott, H. Bögge, and T. Glaser
"Synthesis of Asymmetric Variants in a Dinucleating Ligand Family and Application for Dinuclear Copper(II) Complexes"
Eur. J. Inorg. Chem., 2024, 27, e202400142 - F. Depenbrock, T. Limpke, A. Stammler, J. Oldengott, H. Bögge, and T. Glaser
"Increasing the electron donation in a dinucleating ligand family: molecular and electronic structures in a series of CoIICoII complexes"
Dalton Trans., 2024, 53, 9554-9567
- L. Siebe, C. Butenuth, A. Stammler,H. Bögge, S. Walleck, and T. Glaser
"Generation and Reactivity of µ-1,2-Peroxo CuIICuII and Bis-µ-oxo CuIIICuIII Species and Catalytic Hydroxylation of Benzene to Phenol with Hydrogen Peroxide"
Inorg. Chem., 2024, 63, 2627-2639
- L. Siebe, C. Butenuth, A. Stammler,H. Bögge, S. Walleck, and T. Glaser
"Generation and Reactivity of µ-1,2-Peroxo CuIICuII and Bis-µ-oxo CuIIICuIII Species and Catalytic Hydroxylation of Benzene to Phenol with Hydrogen Peroxide"
Inorg. Chem., 2024, 63, 2627-2639
- S. Finke, A. Stammler, J. Oldengott, S. Walleck, and T. Glaser
"Direct and remote control of electronic structures and redox potentials in µ-diferric complexes"
Dalton Trans., 2023, 52, 17548-17561. - F. Depenbrock, T. Limpke, E. Bill, D. J. SantaLucia, M. van Gastel, S. Walleck, J. Oldengott, A. Stammler, H. Bögge, and T. Glaser
"Reactivities and Electronic Structures of µ-1,2-Peroxo and µ-1,2-Superoxo CoIIICoIII Complexes: Electrophilic Reactivity and O2 Release Induced by Oxidation"
Inorg. Chem., 2023, 62, 17913-17930. -
F. Depenbrock, T. Limpke, A. Stammler, J. Oldengott, H. Bögge, and T. Glaser
"Molecular and Electronic Structures of a Series of Dinuclear CoII Complexes Varied by Exogenous Ligands: Influence of π-Bonding on Redox Potentials"
Eur. J. Inorg. Chem., 2022, e202100992. -
F. L. B. Röhs, S. Dammers, A. Stammler, J. Oldengott, H. Bögge, E. Bill, and T. Glaser
"Dinuclear Diferrous Complexes of a Bis(tetradentate) Dinucleating Ligand: Influence of the Exogenous Ligands on the Molecular and Electronic Structures"
Eur. J. Inorg. Chem., 2022, e202200177. -
S. Walleck, T. P. Zimmermann, H. Hachmeister, C. Pilger, T. Huser, S. Katz, P. Hildebrandt, A. Stammler, H. Bögge, E. Bill, and T. Glaser
"Generation of a µ-1,2-hydroperoxo FeIIIFeIII and a µ-1,2-peroxo FeIIIFeIV Complex"
Nat. Commun., 2022,13, 1376. T. P. Zimmermann, N. Orth, S. Finke, T. Limpke, A. Stammler, H. Bögge, S. Walleck, I. Ivanovic-Burmazovic, and T. Glaser
"Catalytic H2O2 Activation by a Diiron Complex for Methanol Oxidation"
Inorg. Chem., 2020, 59, 15563-15569.S. Walleck and T. Glaser
"A Dinucleating Ligand System with Varying Terminal Donors to Mimic Diiron Active Sites"
Isr. J. Chem., 2020, 60, 1019-1031.-
T. Glaser
"A dinucleating ligand system with varying terminal donor functions but without bridging donor functions: Design, synthesis, and applications for diiron complexes"
Coord. Chem. Rev., 2019, 380, 353-377. T. Zimmermann, S. Dammers, A. Stammler, H. Bögge, and T. Glaser
"Reactivity Differences for the Oxidation of FeIIFeII to FeIII(µ-O)FeIII Complexes Caused by Pyridyl versus 6-Methyl-Pyridyl Ligands"
Eur. J. Inorg. Chem., 2018, DOI: 10.1002/ejic.20180106.M. Aschenbrenner, A. Stammler, H. Bögge, and T. Glaser
"Synthesis and Characterization of a µ-Oxo-Bridged Diferric Complex: An Attempt to Influence the Conformation by Changing the Spacer"
Z. Anorg. Allg. Chem, 2018, 644, 1439-1444.T. P. Zimmermann, T. Limpke, A. Stammler, H. Bögge, S. Walleck, and T. Glaser
"Variation of the Molecular and Electronic Structures of µ-oxo Diferric Complexes with the Bridging Motive"
Z. Anorg. Allg. Chem., 2018, 644, 683-691.T. P. Zimmermann, T. Limpke, N. Orth, A. Franke, A. Stammler, H. Bögge, S. Walleck, I. Ivanovic-Burmazovic, and T. Glaser
"Two Unsupported Termial Hydroxido Ligands in a µ-oxo Ferric Dimer: Protonation and Kinetic Lability Studies"
Inorg. Chem, 2018, 57, 10457-10468.T. P. Zimmermann, T. Limpke, A. Stammler, H. Bögge, S. Walleck, and T. Glaser
"Reversible Carboxylate Shift in a µ-oxo Diferric Complex in Solution by Acid-/Base-Addition"
Inorg. Chem, 2018, 57, 5400-5405.T. Limpke, C. Butenuth, A. Stammler, H. Bögge and T. Glaser
"A Series of Copper Complexes of a Dinucleating Bis(tetradentate) Nitrogen Ligand: Synthesis, Structural, Spectroscopic, Electrochemical, and Magnetic Characterization"
Eur. J. Inorg. Chem, 2017, 29, 3570-3579.S. Dammers, T. P. Zimmermann, S. Walleck, A. Stammler, H. Bögge, E. Bill, and T. Glaser
"A Mixed-Valence Fluorido-Bridged FeII-FeIII Complex"
Inorg. Chem, 2017, 56, 1779-1782.J. B. H. Strautmann, S. Dammers, T. Limpke, J. Parthier, T. P. Zimmermann, S. Walleck, G. Heinze-Brückner, A. Stammler, H. Bögge, and T. Glaser
"Design and Synthesis of a Dinucleating Ligand System with Varying Terminal Donor Functions that Provides no Bridging Donor and its Application to the Synthesis of a Series of FeIII-μ-O-FeIII Complexes"
Dalton Trans., 2016, 45, 3340-3361.G. Heinze-Brückner, S. Walleck, J. B. H. Strautmann, A. Stammler, H. Bögge, and T. Glaser
"Synthesis and Characterization of a Dinuclear Ferric Complex with Redox-active Ligands" "
Inorg. Chim. Acta (Wolfgang Kaim Honorary Issue), 2011, 374, 385-391.J. B. H. Strautmann, S. Walleck, H. Bögge, A. Stammler, and T. Glaser
"A Taylor-made Ligand to Mimic the Active Site of Diiron Enzymes: An Air-Oxidized High-Valent FeIII h.s.(µ-O)2FeIV h.s. Species"
Chem. Commun. 2011, 47, 695-697.J. B. H. Strautmann, C.-G. v. Richthofen, S. DeBeer, E. Bothe, E. Bill, T. Weyhermüller, A. Stammler, H. Bögge, and T. Glaser
"Molecular and Electronic Structures of Dinuclear Iron Complexes Incorporating Strongly Electron-donating Ligands: Implications for the Generation of the One- and Two-Electron Oxidized Forms"
Inorg. Chem. 2011, 50, 155-171.J. B. H. Strautmann, C.-G. Frhr. von Richthofen, S. DeBeer George, E. Bothe, E. Bill, and T. Glaser
"Highly Oxidized Diiron Complexes: Generation, Spectroscopy, and Stabilities"
Chosen as Chem. Comm. Hot Article
Chem. Comm. 2009, 2637-2639.J. B. H. Strautmann, S. DeBeer George, E. Bothe, E. Bill, T. Weyhermüller, A. Stammler, H. Bögge, and T. Glaser
""Molecular and Electronic Structures of Mononuclear Iron Complexes and their Oxidizing Forms Using Strongly Electron-donating Ligands""
Inorg. Chem. 2008, 47, 6804-6824.T. Glaser, R. Pawelke, and M. Heidemeier
"Synthesis, Structure, and Spectroscopic Properties of a Dinuclear Fe(III)-(µ2-O)-Fe(III) Complex Using a Strongly Electron-Donating Ligand: Implications for the Generation of New High-Valent Species"
Z. Anorg. Allg. Chem. (Bernt Krebs Honorary Issue) 2003, 629, 2274-2281.